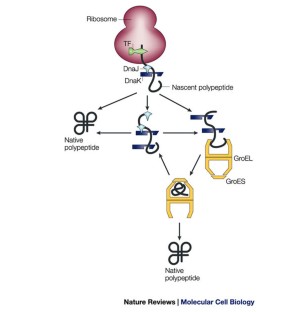
A kinetic partitioning model of selective binding of nonnative proteins by the bacterial chaperone SecB. The binding cascade of SecB to SecA to SecY/E mediates preprotein targeting to the E. U., Lecker, S., Schiebel, E., Hendrick, J. Multitasking SecB chaperones in bacteria. Molecular chaperones are nanomachines that catalytically unfold misfolded and alternatively folded proteins. Molecular chaperones in cellular protein folding: the birth of a field. Chaperone machines for protein folding, unfolding and disaggregation. Molecular chaperone functions in protein folding and proteostasis. Molecular chaperones and protein quality control. The data show how the different architectures of chaperones result in distinct binding modes with non-native proteins that ultimately define the activity of the chaperone.īukau, B., Weissman, J. This unique complex architecture alters the kinetics of protein binding to SecB and confers strong antifolding activity on the chaperone. The multivalent binding mode results in proteins wrapping around SecB. SecB uses long hydrophobic grooves that run around its disk-like shape to recognize and bind to multiple hydrophobic segments across the length of non-native proteins. Here we report the solution structure of SecB, a chaperone that exhibits strong antifolding activity, in complex with alkaline phosphatase and maltose-binding protein captured in their unfolded states. Different chaperones often exert distinct effects, such as acceleration or delay of folding, on client proteins via mechanisms that are poorly understood. Molecular chaperones act on non-native proteins in the cell to prevent their aggregation, premature folding or misfolding.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
